Marty Makary, the current commissioner of the Food and Drug Administration (FDA), has proposed a sweeping change: making nearly all drugs available over the counter (OTC) unless demonstrably unsafe, addictive, or prone to misuse. While framed as deregulation, this plan raises serious safety concerns and highlights existing global trends in drug access.
Expanding OTC Access: What Could Go Wrong?
Makary’s vision – “everything should be over the counter” – is radical, even by pharmaceutical standards. He cites drugs like anti-nausea medicines and vaginal estrogen as examples, but this approach could extend to far more potent substances. The core issue isn’t just availability; it’s that removing prescription requirements eliminates essential oversight, potentially leading to misuse, incorrect dosages, and delayed medical care.
Global Examples: Lessons from Abroad
The U.S. lags behind many countries in OTC access for certain drugs. For example, oral contraceptives containing estrogen and progestin are freely available in over 100 nations, yet remain restricted in the U.S. despite decades of advocacy from the American College of Obstetricians and Gynecologists. They argue wider access reduces unintended pregnancies, but the FDA has resisted, citing safety concerns. Meanwhile, drugs like methocarbamol (Robaxin), a muscle relaxant considered safe elsewhere, are prescription-only in the U.S. This disparity isn’t arbitrary: different countries weigh risk versus access differently.
The Mifepristone Debate: A Case Study in Politics and Safety
The case of mifepristone, used in medical abortions, illustrates the political forces at play. Despite extensive safety data supporting its use, anti-abortion groups have successfully restricted access via the FDA’s Risk Evaluation and Mitigation Strategy (REMS). The drug’s death rate (5 per million users) is lower than that of penicillin (20) or Viagra (49), yet it faces far more scrutiny. Makary’s vow to review its safety, alongside Robert F. Kennedy Jr., suggests further politicization rather than objective assessment.
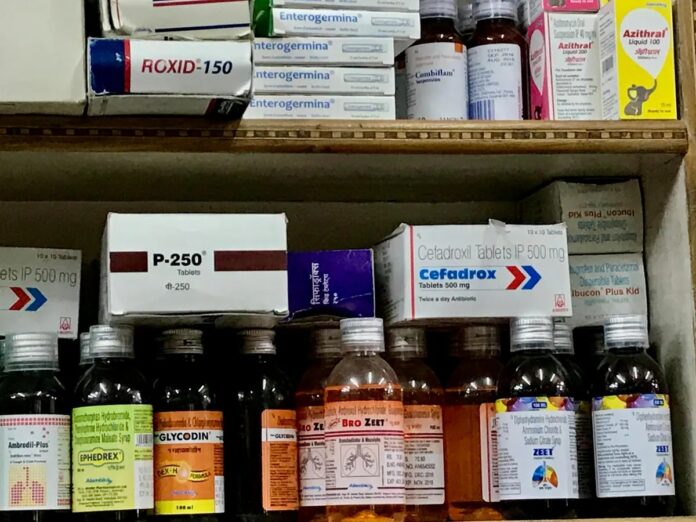
Overlooked Dangers: Common OTC Drugs with Serious Risks
Makary’s focus on controversial drugs ignores the dangers already present in readily available OTC medications. Acetaminophen (Tylenol) overdose can cause fatal liver toxicity. Nonsteroidal anti-inflammatory drugs (NSAIDs) like ibuprofen can lead to severe gastrointestinal bleeding and kidney damage. These risks aren’t theoretical; they’re documented consequences of widespread OTC use. Even President Trump warned against Tylenol during pregnancy, though the science behind his claim is inconclusive, the real danger is untreated fever which is far more dangerous.
The Antibiotic Crisis: A Global Threat Amplified by OTC Access
Perhaps the most alarming implication of Makary’s plan is the potential for unrestricted antibiotic sales. Making antibiotics OTC would accelerate antimicrobial resistance (AMR), a growing global health crisis. Sir Alexander Fleming warned in 1945 about the dangers of self-medication leading to antibiotic-resistant strains. Today, AMR pathogens cause over 2.8 million infections and 35,000 deaths annually in the U.S. alone.
In countries like India and Southeast Asia, where OTC antibiotic use is rampant, drug-resistant bacteria are far more prevalent. A study found 83% of patients in India undergoing a medical procedure carried resistant strains, compared to 20.1% in the U.S. and just 10.8% in the Netherlands.
The Role of Public Health Infrastructure
Addressing the antibiotic crisis requires strengthening public health systems, not dismantling them. The World Health Organization (WHO) advocates for universal health coverage and trusted public health infrastructure to reduce self-medication. However, the U.S.’s withdrawal from the WHO and ongoing budget cuts undermine these efforts. As more Americans become uninsured, reliance on self-medication – including leftover prescriptions or informal sources – will likely increase.
In conclusion, Makary’s push for OTC deregulation isn’t simply about expanding access; it’s about fundamentally altering the landscape of drug regulation. While increased convenience may seem appealing, the potential safety risks and the broader context of global antibiotic resistance paint a far more concerning picture. The U.S. risks repeating mistakes made elsewhere if it prioritizes deregulation over public health.