For decades, antibody therapies have transformed modern medicine, yet they have always faced a fundamental biological barrier: they cannot enter cells. Traditional antibodies are too large to cross the cell membrane, limiting their ability to target diseases that originate inside the cell. However, new research suggests this barrier may soon be breached, offering a promising new avenue for treating cystic fibrosis and potentially other genetic disorders.
By engineering “nanobodies”—tiny, compact fragments of antibodies—scientists have created a therapy that can penetrate cell walls and stabilize faulty proteins from within. This breakthrough not only improves the function of the defective protein responsible for cystic fibrosis but also significantly boosts the effectiveness of existing treatments, bringing patient cells closer to normal health levels.
The Problem with Traditional Antibodies
To understand the significance of this advance, it is necessary to look at the limitations of current antibody drugs. While highly effective for targeting surface receptors or circulating pathogens, full-sized antibodies are physically unable to pass through the lipid bilayer of cell membranes. Consequently, they can only interact with targets on the outside of cells.
Cystic fibrosis, however, is an intracellular problem. It is caused by a mutation in the CFTR gene, which produces a misfolded protein that fails to reach the cell surface. Current treatments use small-molecule drugs to help this protein fold correctly or function better, but these therapies rarely restore the protein to its full, natural capacity. The disease process begins inside the cell, yet traditional antibody technology has been unable to reach it.
Enter the Nanobody
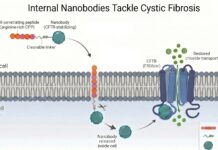
The solution lies in nanobodies. Derived originally from camelids (such as camels and llamas), these are single-domain antibody fragments that are significantly smaller and more stable than conventional antibodies. Their compact size allows them to penetrate tissues more effectively, but size alone was not enough to cross the cell membrane.
Researchers have now solved the delivery problem by fusing the nanobody with a cell-penetrating peptide. This peptide consists of ten positively charged arginine amino acids. Because cell membranes carry a negative charge, the peptide interacts with the cell surface, facilitating uptake into the cell’s interior. Once inside, the nanobody is released to seek out its specific target.
Key Insight: Without this molecular “key” (the peptide), the nanobody would remain outside the cell, useless for intracellular targets. This fusion creates a hybrid molecule capable of navigating the cellular barrier.
Stabilizing the Broken Protein
In patients with cystic fibrosis, the mutated CFTR protein is unstable and misshapen. It gets trapped inside the cell and degraded before it can reach the surface to regulate salt and water transport. This leads to the characteristic thick, sticky mucus that clogs the lungs.
The newly designed nanobody binds directly to the unstable region of the CFTR protein. By attaching itself, the nanobody acts as a structural scaffold, stabilizing the misfolded protein. This stabilization allows the protein to mature correctly, escape degradation, and travel to the cell membrane. Once there, it resumes its role as a functional chloride channel, facilitating proper fluid balance.
Synergy with Existing Therapies
Perhaps the most compelling finding of this research is how this new approach complements existing treatments. Current approved drugs for cystic fibrosis stabilize different regions of the CFTR protein. The nanobody targets a distinct domain affected by the mutation.
When used in combination, these therapies reinforce each other:
- Enhanced Activity: In cell models, pairing the nanobody with standard drug combinations resulted in a substantial increase in CFTR activity compared to either treatment alone.
- Near-Normal Function: In airway cells derived from patients, this combined therapy restored protein function to nearly 90% of normal levels. This is a significant leap forward, suggesting that patients could experience dramatically improved lung health and reduced mucus buildup.
Beyond Cystic Fibrosis
While the immediate focus is on cystic fibrosis, the implications of this technology extend far beyond this single disease. The ability to deliver functional protein drugs inside cells opens the door to treating a wide range of conditions where intracellular proteins malfunction—from rare genetic disorders to common diseases with complex internal mechanisms.
Previous attempts to deliver proteins into cells often relied on gene therapy, which involves inserting genetic material into cells to force them to produce the therapeutic protein. This approach carries its own risks and complexities. The nanobody method, by contrast, delivers the protein directly without altering the patient’s DNA, offering a potentially safer and more straightforward path to treatment.
Challenges and Future Directions
Despite the promise, hurdles remain. The primary challenge is delivery in complex environments like the human lung, where thick mucus can block access to target cells. Further research is needed to refine how these nanobodies are administered to ensure they reach the right cells in sufficient quantities.
However, the proof of concept is now clear: protein drugs can be engineered to cross cellular barriers and correct disease mechanisms at their source. This breakthrough expands the horizon of what antibody-based therapies can achieve, moving medicine from the cell surface to the cell interior.
In summary, this innovation represents a paradigm shift in drug design. By enabling nanobodies to enter cells and stabilize faulty proteins, scientists have unlocked a new level of precision in treating genetic diseases, offering hope for more effective and comprehensive therapies in the near future.